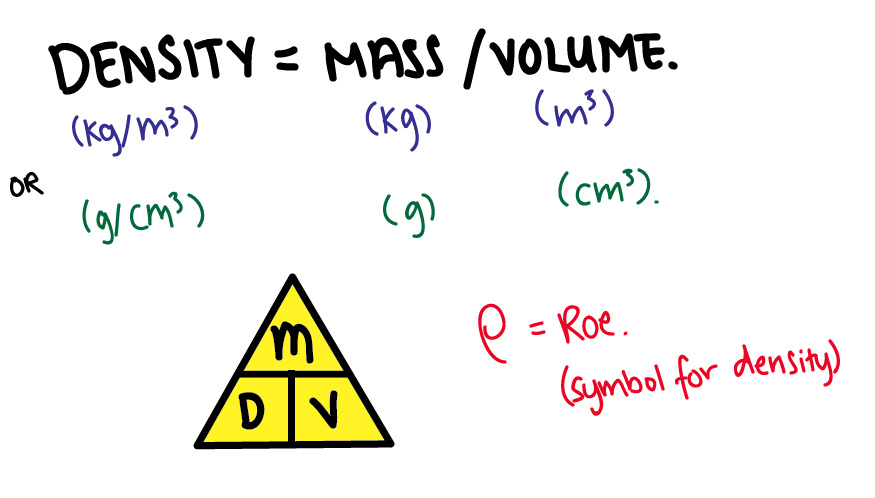The Three States of Matter
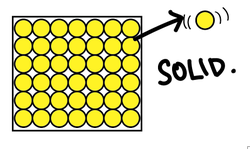
In solids, the particles are closely packed in a regular (lattice) arrangement.
The particles vibrate, held in place by strong, springlike forces between particles.
The particles vibrate, held in place by strong, springlike forces between particles.
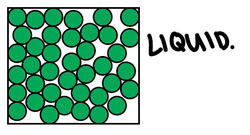
In liquids, the particles are still closely packed, but they have no fixed/regular arrangement - they are randomly arranged.
There are strong forces of attraction between the particles, and they move randomly but slide past each other due to limited space.
There are strong forces of attraction between the particles, and they move randomly but slide past each other due to limited space.
In gases, the particles are spread out - there are large spaces between the randomly arranged particles.
The particles are free to move, and do so at high speeds. They bounce off each other and the walls, exerting a force.
In an ideal gas, there are zero forces between the particles.
The particles are free to move, and do so at high speeds. They bounce off each other and the walls, exerting a force.
In an ideal gas, there are zero forces between the particles.
Particles in all states of matter are constantly moving - this is called the Kinetic Theory.
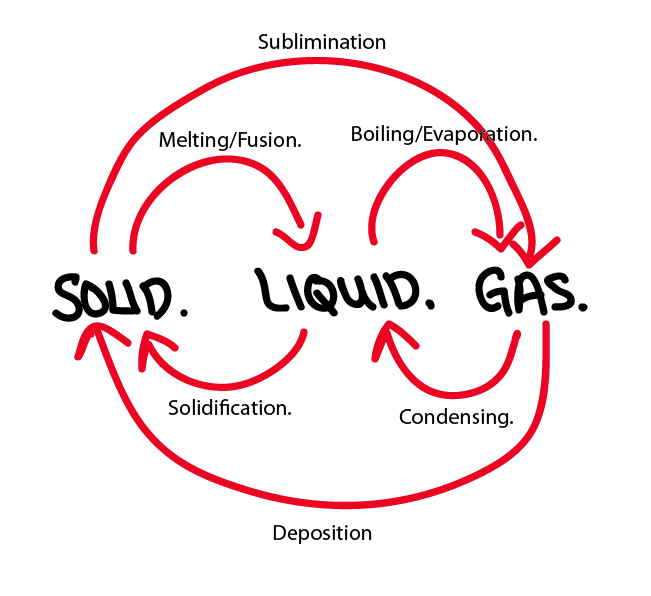
Changing State.
Melting:
The change of state where a solid becomes a liquid at the melting point. The melting point is the temperature at which a solid melts.
Boiling:
The change of state where a liquid becomes a gas at the boiling point. The boiling point is the temperature at which the solid melts.
Evaporation:
The change of state where a liquid becomes a gas at a temperature below the boiling point.
The change of state where a solid becomes a liquid at the melting point. The melting point is the temperature at which a solid melts.
Boiling:
The change of state where a liquid becomes a gas at the boiling point. The boiling point is the temperature at which the solid melts.
Evaporation:
The change of state where a liquid becomes a gas at a temperature below the boiling point.